Summary
Background: Pneumomediastinum is defined as the presence of air in the mediastinal region. It is associated with events or diseases leading to a sudden increase in alveolar pressure. The air in the mediastinum may originate in the pharynx, the tracheobronchial tree or the esophagus. This study aimed to assess the clinical and radiological characteristics of patients who were diagnosed with spontaneous pneumomediastinum and received treatment at our clinic.Materials and Methods: A total of 23 patients who had spontaneous pneumomediastinum and were treated at our clinic between 2007 and 2019 were retrospectively assessed for their age, etiological factors, clinical and radiological characteristics; treatment and outcomes. Chest radiography and computed tomography of the thorax were used for diagnosis in all patients.
Results: Of the patients, 15 were males and eight females and their mean age was 33.91 (15-82) years. The most common symptom on admission was shortness of breath (n = 12) and chest pain (n = 11). The etiological factors were excessive vomiting in six patients and excessive cough in five. Radiologically, the most common findings besides pneumomediastinum were subcutaneous emphysema (n = 6) and pneumothorax (n = 4). Of the patients, seven required esophagoscopy, four required bronchoscopy and no pathology was observed. All patients were fully recovered when they were discharged.
Conclusions: The underlying etiological factor is crucial in pneumomediastinum. Conservative treatment methods are often sufficient in spontaneous pneumomediastinum patients with no pathology in endoscopic evaluation.
Introduction
Pneumomediastinum (mediastinal emphysema) is defined as the presence of air in the mediastinum observed on chest radiography or computed tomography (CT) of the thorax. Spontaneous pneumomediastinum (SPM), a very rare clinical condition, is defined as nontraumatic pneumomediastinum without surgical or endoscopic interventions, thoracic trauma or mechanical ventilation support [1-3]. SPM was first reported by Hamman in 1939, with an incidence range from 1:7000 to 1:45000 in all hospital admissions [4-6].In many patients, pneumomediastinum is associated with events or diseases causing a sudden increase in the alveolar pressure. It causes alveolar rupture through an increase in alveolar pressure and bronchial obstruction of the check-valve type. Due to this alveolar rupture, the air in the lung parenchyma proceeds towards the interstitial region around the vessel and the bronchus, then towards the lung hilus and the mediastinum. The air goes through the mediastinum, anterior chest wall and between the large vessels in the neck, which leads to subcutaneous emphysema. It may also go inside the visceral pleura through the peribronchial and vascular structures and cause pneumothorax if the visceral pleura is ruptured [2,3].
Many factors are involved in the etiology of spontaneous pneumomediastinum. Deep breathing maneuvers such as forced vital capacity or excessive exercise, Valsalva maneuvers such as giving birth or weight-lifting, closed chest trauma, especially in patients with obstructive pulmonary disease and who receive positive end-expiratory pressure therapy and the sudden atmospheric pressure drops such as in a rapid descent by a diver or pilot are among the known etiological causes. Of the cases, 30% present with no etiological cause [5-7].
In this study, we aimed to retrospectively assess the clinical and radiological characteristics of patients who had spontaneous pneumomediastinum and were treated at our clinic.
Methods
Twenty-three patients treated with the diagnosis of SPM in our clinic between the years 2007-2019 were assessed retrospectively. The patients were evaluated according to their age, gender, etiological factors, smoking habits, clinical and radiological characteristics and treatment outcomes. After the diagnosis of SPM, oral intake was discontinued for 24 hours, and nasal oxygen therapy, bed rest, prophylactic antibiotics (cefazolin sodium 1000 mg, 2x1 iv.) and pain relief treatment (diclofenac 75 mg/3 mL, im.) were provided when necessary. Oral intake was re-initiated and prophylactic antibiotics were discontinued in patients with no pathology (pneumothorax, pleural fluid, mediastinal enlargement and parenchymal infiltration) in their chest radiography at the 24th and 48th hours of hospitalization and with normal laboratory values (Hb, Hct and leukocyte). Patients with normal radiological and clinical findings were discharged on the following day. The study was approved by the institutional review board (No: 2020/654) and conducted in accordance with the principles of the Declaration of Helsinki.Results
Of the 23 patients diagnosed with spontaneous pneumomediastinum, 15 were males and eight females. Their mean age was 33.91 (15-82) years. Symptoms on admission were shortness of breath in 12 patients, chest pain in 11, difficulty swallowing in five and swelling in the neck and face in five. Of them, 10 had accompanying diseases (COPD in five, malignancy in three) and 14 (61%) had a history of smoking (TableTable 1: Clinical and radiological characteristics of the patients.
Considering the underlying etiologies of our patients, six (26%) had excessive retching or vomiting and five (21%) had an excessive cough. All cases underwent CT of the thorax and six of them had subcutaneous emphysema, four had pneumothorax and three had neck emphysema. The CT findings of two patients raised suspicion of esophageal rupture, but no rupture was detected in rigid esophagoscopy for diagnostic purposes.
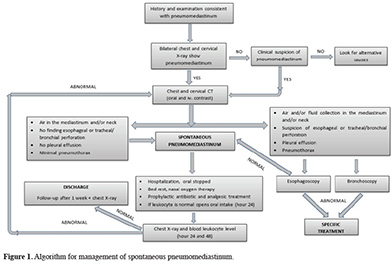
Esophagoscopy was performed as an invasive procedure in seven patients, especially in four of them with excessive vomiting, and bronchoscopy was done in four. No esophageal or bronchial injury was detected in any of these endoscopic interventions. Two patients required VATS wedge resection for treatment purposes. All the cases had SPM and all were fully recovered when they were discharged. Morbidity was observed in two cases, one due to pneumonia caused by excessive vomiting and one due to prolonged air leakage following VATS bullous wedge resection. Mean duration of hospitalization was 4.43 (2-14) days and no problems were observed in any of our patients in their outpatient follow-ups. Figure 1 shows the algorithm for the diagnosis and treatment of spontaneous pneumomediastinum, created in accordance with our results.
 Click Here to Zoom |
Figure 1: Algorithm for management of spontaneous pneumomediastinum. |
Discussion
Spontaneous pneumomediastinum is a rare clinical condition, which is often observed in tall young men and usually heals spontaneously. Bronchial asthma is the most common etiological factor accompanying SPM (also known as nontraumatic pneumomediastinum) [8]. Pneumomediastinum has an incidence of 1:7000 to 1:45000 per year among all hospital admissions in adults and of 2.5/1000 per year in all live births [4-6]. SPM was detected in 23 (0.48%) of nearly 4710 patients consulted to our clinic in the emergency department during the study period.Spontaneous pneumomediastinum develops after physical activities that increase intrathoracic pressure such as excessive cough, intense exercise, Valsalva maneuvers; an excessive strain of the esophagus and vaginal delivery. Moreover, barotrauma, the snorting of cocaine and other drugs can increase alveolar and intrabronchial pressure and lead to pneumomediastinum and subcutaneous emphysema. There have also been rare cases reported in the literature including pulmonary function testing, playing wind instruments, inflating balloons and convulsion [1-3]. A severe cough in children caused by pertussis, diphtheria, bronchiolitis or acute bronchitis can also cause pneumomediastinum [9,10]. Mihos et al. reported some causes of pneumomediastinum to be doing sports, scuba diving and playing basketball and football [11]. Of the cases, 39% to 100% present with a precipitating factor [3,8]. In accordance with the literature, 65% our patients had an underlying precipitating factor and the most common of them were vomiting and excessive cough.
Of the SPM cases, 32% are active smokers and 14% to 18% have bronchial asthma or emphysema [3,7,8]. We found that 60.8% of our patients were active smokers and 22% had bronchial asthma or emphysema as an accompanying disease.
In pneumomediastinum, if the mediastinum pressure rises suddenly or decompression fails to be sufficient, the mediastinal pleura ruptures and causes pneumothorax which can be observed in around 10-18% of the SPM cases [2,3]. The tension pneumomediastinum is the more severe clinical form of pneumomediastinum which mimics cardiac tamponade [7]. Pneumothorax was detected in four (17.4%) of our patients, while none had tension pneumomediastinum. None of the patients diagnosed with pneumothorax required tube thoracostomy and all the patients were recovered by nasal oxygen therapy.
The clinical presentation involves sudden chest pain accompanying shortness of breath, cyanosis, fullness in the neck veins; feeling of something being stuck in the throat, dysphagia, dysphonia and neck and subcutaneous emphysema. The signs and symptoms depend largely on the amount of air in the mediastinal space and the presence of a concomitant infection [1,7]. In the clinic, localized retrosternal chest pain is the most common symptom. The pain starts suddenly and increases with deep breaths and coughs [3]. In our patients, the most common symptoms were shortness of breath at 52% and chest pain at 48%, followed by difficult swallowing. In fact, the amount of air in the mediastinum is the only cause of all these findings and the symptoms of a rupture of the mediastinal pleura due to the amount of air in the mediastinum determine the patient’s clinic.
Physical examination often indicates the presence of air in the subcutaneous tissues in the neck and chest wall [7,8]. The typical physical finding of SPM is a cracking sound (Hamman’s Sign) at auscultation in the anterior chest, synchronous with peak heart rate. Hamman’s Sign is a typical finding for physical examination, although it’s detected only in 10-20% of the patients [1-3]. The most common finding of physical examination was subcutaneous and neck emphysema in our patients and Hamman’s Sign was detected in none.
PA chest radiography and thorax CT have an important role in the diagnosis of SPM. On PA chest radiography, accumulation of air may be detected surrounding the mediastinal pleura, esophagus or the main bronchi. Thorax CT is the most sensitive method to diagnose pneumomediastinum and should be preferred, particularly in cases with no determined underlying etiological factor. Observing an air density in the mediastinum on thorax CT confirms the diagnosis [7,8,12]. We use thorax CT in the radiological diagnosis of SPM at our clinic and demand a CT with oral and intravenous contrast if there is suspicion of esophageal perforation in the patient’s history. The most common radiological findings on thorax CT were subcutaneous or neck emphysema and pneumothorax in our patients. Two of them were suspected for esophageal perforation on radiology, however, no pathology was observed in the procedure for endoscopic diagnosis.
The differential diagnosis of spontaneous pneumomediastinum includes acute coronary syndrome, pericarditis, pneumothorax; pulmonary embolism, tracheobronchial rupture and Boerhaave Syndrome [1-3]. Following the diagnosis of pneumomediastinum, bronchoscopy and esophagoscopy should be performed if the patient is suspected for rupture of the airways or the esophagus. The patient’s clinic and thorax CT findings are key in guiding the endoscopy decision. Esophagoscopy was performed in seven of our patients and bronchoscopy in four and no pathology was detected.
The treatment approach should mostly be conservative (bed rest, pain relief, prophylactic antibiotics and nasal oxygen therapy) if there is no injury in the main bronchus or the esophagus in the treatment of pneumomediastinum and no large source of air leakage can be detected from lung tissue (such as bulla or bleb). Nasal oxygen support and bed rest are the most important elements in treatment and antibiotics are recommended for mediastinitis prophylaxis [2,3].
The aim is to make sure not to overlook a serious underlying cause leading to this condition in the management of pneumomediastinum. These patients should be followed-up for at least 24-36 hours. This clinical condition often regresses spontaneously within 48 hours. Oral administration is initiated and the patients are discharged with follow-up including chest radiography and complete blood count. Mihos et al. reported a mean hospital stay of 3.8 days in their 25 cases with pneumomediastinum due to sports injuries [11]. Our patients had a mean follow-up period of 4.4 days. The recurrence rate of untreated spontaneous pneumomediastinum is quite rare. Abolnik et al. reported recurrence in two of 25 patients over a two-year period [13]. We observed no recurrence in our patients.
SPM is often a benign and restricting clinical condition with no mortality. Mortality rates reach up to 40% in secondary pneumomediastinum while this rate is 0% in spontaneous pneumomediastinum [7,8]. Morbidity was observed in two patients, however, mortality was observed in none in our series.
To conclude, spontaneous pneumomediastinum is a benign clinical condition which needs to be followed-up by conservative treatment methods. If an injury of the airways or esophagus is considered in the etiology, endoscopic intervention with cause-based treatment should be performed. Although it is rarely observed in healthy adolescents and young adults, spontaneous pneumomediastinum should be considered in the differential diagnosis when sudden chest pain and shortness of breath are present.
Declaration of conflicting interests
The authors declared no conflicts of interest with respect to the authorship and/or publication of this article.
Funding
The authors received no financial support for the research and/or authorship of this article.
Ethics approval
The study was approved by the Ethics Committee of Ondokuz Mayis University (No: 2020/654).
Authors’ contributions
BÇ; Conceived and designed the analysis, collected the data, contributed data/analysis tools, performed the analysis, co-wrote the paper MGP; Conceived and designed the analysis, performed the analysis, VY; Collected the data, SG; Collected the data, co-wrote the paper, YBB; Contributed data/analysis tools, ATŞ, AB; co-wrote the paper.
Reference
1) Takada K, Matsumoto S, Hiramatsu T, Kojima E, Shizu M, Okachi S et al. Spontaneous pneumomediastinum: an algorithm for diagnosis and management. Ther Adv Respir Dis 2009; 3: 301-7.
2) Kim KS, Jeon HW, Moon Y, Kim YD, Ahn MI, Park JK et al. Clinical experience of spontaneous pneumomediastinum: diagnosis and treatment. J Thorac Dis 2015; 7: 1817-24.
3) Sahni S, Verma S, Grullon J, Esquire A, Patel P, Talwar A. Spontaneous pneumomediastinum: time for consensus. N Am J Med Sci 2013; 5: 460-4.
4) Hamman L. Spontaneous mediastinal emphysema. Bull Johns Hopkins Hosp 1939; 64: 1-21.
5) Macia I, Moya J, Ramos R, Morera R, Escobar I, Saumench J et al. Spontaneous pneumomediastinum: 41 cases. Eur J Cardiothorac Surg 2007; 31: 1110-4.
6) Iyer VN, Joshi AY, Ryu JH. Spontaneous pneumomediastinum: analysis of 62 consecutive adult patients. Mayo Clin Proc 2009; 84: 417-21.
7) Caceres M, Ali SZ, Braud R, Weiman D, Garrett HE. Spontaneous pneumomediastinum: a comparative study and review of the literature. Ann Thorac Surg 2008; 86: 962-6.
8) Dajer-Fadel WL, Arguero-Sanchez R, Ibarra-Perez C, Navarro-Reynoso FP. Systematic review of spontaneous pneumomediastinum: A survey of 22 years’ data. Asian Cardiovasc Thorac Ann 2014; 22: 997-1002.
9) Gasser CR, Pellaton R, Rochat CP. Pediatric Spontaneous Pneumomediastinum: Narrative Literature Review. Pediatr Emerg Care 2017; 33: 370-4.
10) Chalumeau, M, Le Clainche L, Sayeg N, Sannier N, Michel JL, Marianowski R et al. Spontaneous pneumomediastinum in children. Pediatr Pulmonol 2001; 31: 67-75.
11) Mihos P, Potaris K, Gakidis I, Mazaris E, Sarras E, Kontos Z. Sports-related spontaneous pneumomediastinum. Ann Thorac Surg 2004; 78: 983-6.






