2Department of Chest Diseases, Acıbadem Kayseri Hospital, Kayseri, Turkey DOI : 10.26663/cts.2021.0026
Summary
Bronchial atresia is one of the rare congenital bronchopulmonary malformations. The affected bronchus is atresic and usually the upper lobe of the left lung is involved and is not connected to the central airways. In this case report, we present the treatment process of a patient with post-intubation tracheal stenosis, left main bronchial stenosis, and an agenesis of left upper lobe bronchus.Introduction
Bronchial atresia is one of the rare congenital bronchopulmonary malformations. Usually, the upper lobe of the left lung is involved and is not connected to the central airways. The affected bronchial system gets filled with mucocele. Atelectasis, hyperlucency, and hypervascularity are observed on CT of the affected lung [1-3]. In this case report, we would like to present the treatment process of a patient with post-intubation tracheal stenosis, left main bronchial stenosis, and an agenesis left upper lobe bronchus.Case Presentation
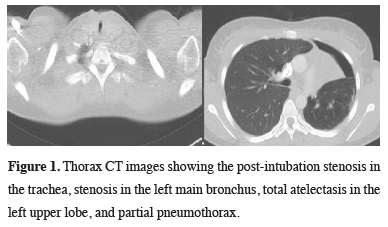
A 33-year-old female patient was admitted to our clinic with a pre-diagnosis of tracheal stenosis and left lung mass. The patient had been hospitalized in an intensive care unit for nearly a year and was treated for tuberculosis. However, no laboratory data of tuberculosis were found in the patient’s file examination. Tracheostomy and bronchoscopy had been performed upon detection of a mass in the upper lobe of the left lung in the examinations of the patient who had developed respiratory distress. Bronchoscopy had showed stenosis in the trachea and a mass lesion that obstructed the left main bronchus almost completely. Biopsy had been performed. No malignancy had been found as a result of biopsy and tuberculosis examination.When the patient was admitted, she had severe stridor and orthopnea. Thorax CT revealed stenosis in the trachea, total obstruction of the left main bronchus, left upper lobe atelectasis, and adjacent partial pneumothorax (Figure 1).
 Click Here to Zoom |
Figure 1: Thorax CT images showing the post-intubation stenosis in the trachea, stenosis in the left main bronchus, total atelectasis in the left upper lobe, and partial pneumothorax. |
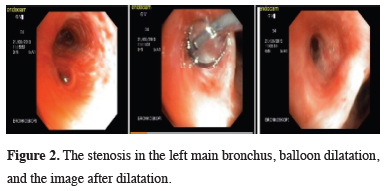
PET-CT was performed on the patient due to suspicion of malignancy. No abnormal FDG uptake was observed in PET-CT. The stenosis in the trachea area was dilated with rigid bronchoscopy. There was a 4 mm opening through which distal left main bronchus could not be reached. It was dilated by performing balloon dilatation in the left main bronchus. Then, when the distal was reached with bronchoscope, it was observed that there was no left upper lobe bronchus. Biopsies were taken from the left main bronchus and the pathology test result was negative again for malignancy (Figure 2).
 Click Here to Zoom |
Figure 2: The stenosis in the left main bronchus, balloon dilatation, and the image after dilatation. |
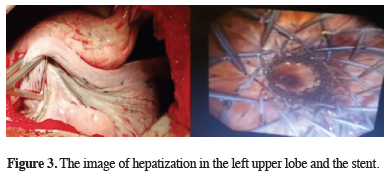
Then, in different sessions, rigid bronchoscopy and balloon dilatations were performed on the patient for 4 more times, and the trachea and left main bronchus were dilated. The left upper lobe bronchial orifice was never observed, and it was decided to perform left upper lobectomy on the patient since it was considered to be atretic. During lobectomy, it was observed that the left upper lobe bronchus was atresic and in the form of a band, just as it was observed in bronchoscopy. A mucocele-like gelatinized liquid was observed to flow through the removed left upper lobe. After thoracotomy, rigid bronchoscopy was performed on the patient again and a stent was placed in the trachea and left main bronchus in the same session (Figure 3). No malignancy was found in the pathological examination of the left upper lobe. The result showed alveolar proteinosis.
 Click Here to Zoom |
Figure 3: The image of hepatization in the left upper lobe and the stent. |
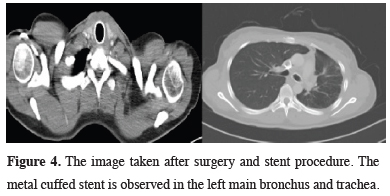
After a post-op follow-up for about 12 months with stent, the stents were removed and the patient has no problem as of the 24th month follow-up at present (Figure 4).
 Click Here to Zoom |
Figure 4: The image taken after surgery and stent procedure. The metal cuffed stent is observed in the left main bronchus and trachea. |
Written informed consent was obtained from the patient for publication of her data.
Discussion
Most tracheal stenosis cases are due to intubation, tracheostomy, and chemical or traumatic injuries. It has been reported that it may be due to other inflammatory diseases such as Wegener granulomatosis, connective tissue diseases, sarcoidosis, and tuberculosis. Idiopathic subglottic stenosis commonly seen in women is among the rare causes of tracheal stenosis [1].Bronchial stenosis together with tracheal stenosis is usually seen in childhood and it is congenital. It is often seen together with additional deformities such as pulmonary artery anomaly, ventricular septal defect, lung hypoplasia, lung agenesis, musculoskeletal anomalies, central nervous system anomalies, genitourinary system anomalies, and gastrointestinal defects [2-4]. There are publications stating that congenital bronchial atresia is the result of bronchial artery occlusion that develops during the fetal period [5]. In this case, there was atresia in the left upper lobe bronchus, stenosis in the left main bronchus, and post-intubation tracheal stenosis. In the history of our patient, there was a treatment in intensive care unit with a pre-diagnosis of tuberculosis for a period of approximately one year. However, no evidence of tuberculosis was found in any specimen taken after the operation. In addition, none of the other anomalies accompanied by bronchial stenosis and agenesis were seen in our patient.
Congenital bronchial atresia was first reported by Ramsay in 1953 [6]. Pulmonary agenesis is morphologically categorized according to the presence or absence of bronchial, alveolar, and vascular structures and whether their agenesis is bilateral or unilateral [3]. While the absence of bronchus, lung parenchyma, and vascular structures together is called pulmonary agenesis, the absence of lung parenchyma with rudimentary bronchus is called aplasia, and the presence of a rudimentary lung tissue with rudimentary bronchus is called pulmonary hypoplasia [7,8]. In our case, besides left main bronchial stenosis, endoscopic and postoperative evaluation did not show a luminal left upper lobe bronchus, but there was an atelectasis lung tissue. As such, it does not comply with the description of the current classifications.
In the evaluation of bronchial malformations, thorax CT angiography and bronchoscopy are the basic diagnostic tests. Sometimes, accompanying cardiac anomalies and anatomical anomalies of pulmonary vascular structures can be clearly identified with angiography performed with cardiac catheterization [3]. In bronchial atresia, air retention due to collateral circulation and a cystic area (bronchocele) distal to the obstructed bronchus may be observed in tomography. Sometimes the parenchyma cannot be observed in cases where the lobe bronchus and parenchyma structure did not develop at all, or it may be the absence of a sequestered parenchymal structure or bronchial and vascular structures [9]. It has been reported that bronchial atresia is mostly seen in the left upper lobe and especially the segmental ones are seen in the apicoposterior segment. PET-CT was performed in one patient with a suspicion of mass and no pathological FDG uptake was observed. It was stated that the appearance of mass in the patient was due to the mucocele accumulated in the ectatic bronchus in the diaphragm of the atresia bronchus [10]. Common long-term complications seen in bronchial atresia are pneumothorax, recurrent pneumonia, and pulmonary parenchymal deterioration [9]. In this case, the right upper lobe was in total atelectasis. There was a partial pneumothorax adjacent to the atelectasic upper lobe and no pathological FDG uptake was observed on PET-CT due to the appearance of the mass. In our case, the inside of the removed upper lobe was completely filled with mucus accumulation, and a result of alveolar proteinosis was reported after the pathological examination.
Among the causes of acquired bronchial stenosis, reasons such as selective main bronchial intubation, repeated intrathoracic vascular interventions, and cardiovascular stent compression have been stated [2]. In the literature, a patient who developed stricture completely occluding the right main bronchus despite treatment after tuberculosis has been presented. It is stated that with serial balloon dilatations, the total atelectasis of the patient’s right lung was opened and the clinical course improved rapidly [11]. Andrews stated that after tuberculosis, the left bronchial system was totally obstructed by fibrosis due to tuberculosis in one of two cases, and in the other case, the right upper lobe bronchus was obstructed by tuberculosis-related fibrosis [12]. Since this case had a history of tuberculosis treatment in the past, it was the first etiology that came to mind that there might be atresia developing after tuberculosis, however, no microbiological evidence of tuberculosis was found in the past file examination of our case, and no data on tuberculosis was found as a result of the pathological examination of the specimens we obtained.
In conclusion, bronchial atresia is a rare condition. The coexistence of bronchial atresia and stenosis in the trachea and bronchi has not been encountered in the literature. In this case, it is interesting that both lobar bronchial atresia, tracheal stenosis, and stenosis in the left main bronchus were together, and there was no evidence of anomaly in any other accompanying organs and vascular structures in terms of etiopathology. We present this case since the present findings are different from the cases with bronchial anomalies discussed in the literature.
Declaration of conflicting interests
The authors declared no conflicts of interest with respect to the authorship and/or publication of this article.
Funding
The authors received no financial support.
Authors’ contributions
BM, MT: conceived and designed the current case report, co-wrote the paper, collected the clinical data. The authors discussed the case under the literature data together and constituted the final manuscript.
Reference
1) Aydoğmuş Ü, Yuncu G, Türk F. A case of idiopathic subglottic and bilateral bronchial stenosis. Turk Thorac J 2016; 7: 28-31.
2) Schweiger C, Cohen AP, Rutter MJ. Tracheal and bronchial stenoses and other obstructive conditions. J Thorac Dis 2016; 8: 3369-78.
3) Joo CU, Song GY, Kim JS. Lobar agenesis of the left upper lung. J Korean Med Sci 1990; 5: 233-5.
4) Al-Qadi MO, Reddy DRS, Larsen BT, Iyer VN. Chronic pulmonary aspergillosis complicating bronchial atresia. Case Rep Med 2014; doi: 10.1155/2014/208963.
5) Murata S, Oguri T, Sasada S, Tsuchiya Y, Ishioka K, Takahashi S et al. A case of congenital bronchial atresia patient with subclinical infection who underwent lung resection. Resp Med Case Rep 2020; 30: 101076.
6) Ramsay BH. Mucocele of the lung due to congenital obstruction of a segmental bronchus. Dis Chest 1953; 24: 96-103.
7) Ghaye B, Szapire D, Fanchamps JM, Dondelinger RF. Congenital bronchial abnormalities revisited. Radiographics 2001; 21: 105-19.
8) Gupta K, Taneja D, Aggarwal M, Gupta R. Left upper lobar agenesis of lung: a rare case report. Lung India 2017; 34: 302-4.
9) Traibi A, Seguin-Givelet A, Grigoroiu M, Brain E, Gossot D. Congenital bronchial atresia in adults: thoracoscopic resection. J Vis Surg 2017; 3: 174.
10) Demir M, Taylan M, Yılmaz S, Dursun E, Sezgi C, Işık R. Bronchial atresia in an adult misdiagnosed as pulmonary mass. Tuberk Toraks 2017; 65: 351-3.
11) Tan GP, Abisheganaden JA, Goh SK, Verma A. Reversible platypnoea-orthodeoxia syndrome in post-tuberculosis bronchial stenosis. Respirol Case Rep 2018; 6: e00303.
12) Andrews CH. Bronchial stenosis in pulmonary tuberculosis. Can Med Assoc J 1935; 33: 36-41.






