Summary
Primary pulmonary sarcomas of the lung are rare tumors with a low incidence. We reviewed the literature and analyzed 5 patients retrospectively. All clinical datas and therapeutic protocols were recorded. The required surgical procedure was lobectomy in 4 patients and diagnostic broncoscopy and mediastinoscopy in one patient. At histological examination; 2 carcinosarcoma, 1 undifferentiated pleomorphic sarcoma, 1 leiomyosarcoma and 1 synovial sarcoma diagnosed. Primary sarcomas of the lung cannot be differentieted from more common tumors of the lung tumors clinically. Early diagnosis and complete surgical resection is critical for prognosis.Introduction
Primary pulmonary sarcomas of the lung are very rare malignant tumors of the lung [1]. The incidence of this tumor is very low. They account less then 0.5% of all lung tumors and whe have very limited knowledge about the clinical behaviour and futures when compared to the other lung tumors [2].Differantial diagnosis is critical for treatment. Additional histopathological and immunhistochemistery (IHC) examination could be required for a definitive diagnosis [1,2].
Surgical management consists radical resection with pathological free margins is still the gold standart for the treatment. Postoperatively chemotherapy and/or radiotherapy is often advised. However, a standart therapy protocol is not clear for the patients having primary pulmonary sarcoma of the lung [1,2].
We retrospectively analyzed 5 patients with primary sarcoma of the lung managed in our department during a 5-year-period in a single instution and reviewed the international medical literature.
Case Presentation
Records of 5 patients diagnosed with primary pulmonary sarcoma of the lung between 2014 and 2018 in our department were reviewed. Demographic data, past medical and familial history, and all clinical symptoms were noted. Chest films, computed tomography (CT) and positron-emission tomography (PET) scans were examined for the tumor location, tumor size, invasion, metastasis and maximum standart uptake value (SUV-max). Therapeutic protocols of surgery, chemotherapy and/or radiotherapy were also recorded. Pathological slides were examined by the same pathologist, experienced in the lung tumors. Electron microscopy and IHC were also performed. The mean diameter of the tumor, invasion (lymph nodes, pleura, diaphragma, muscle, chest wall, etc.), surgical margins and pathologic diagnosis were recorded. Follow-up period of the patients after surgery were listed. Detailed clinical and evaluative features of all patients are given at table 1.Table 1: Detailed clinical and evolutive features of patients
Case 1
A 58-year-old man with severe right lateral chest pain was admitted to our clinic. He was a heavy smoker and his medical history is nonspesific. Blood chemistry and hematological examinations were normal. A chest X-ray and CT showed a 7.5 cm solid lesion at right upper lobe. At PET/CT scan, SUV-max of the mass was 9.2 g/ml and there were no lymph node invasion or distant metastasis. The patient was operated and right upper lobectomy and mediastinal lymph node disection was performed. At pathological and IHC examination carcinosarcoma diagnosed, the mean diamater of tumor was 7.5x7x5.5 cm, and there were no lymph node or pleura invasion with free margins. After the surgical procedure patient has received chemotherapy and radiotherapy. He is still alive and follow up from 52 months without recurrence and metastasis.
Case 2
A 65-year-old man previously healthy man admitted to our clinic for a 2 cm lesion of left upper lob. The patient was asymptomatic and the mass was diagnosed incidentally during a check up. CT and PET/CT revealed a 2 cm peripheral cavitary lesion at the left upper lobe (SUV-max 11 g/ml). A left upper lobectomy was performed, at pathological and IHC examination, undifferentiated pleomorphic sarcoma was diagnosed. Diameter of tumor was 2.1 cm and there were no lymph node invasion. Chemotherapy and/or radiotherapy was not performed after the surgery. The patient is free of disease and followed up for 33 months.
Case 3
A 52-year-old women presented with dyspnea and hemoptysis for 2 months. She also had a history of hyperthyroidism and underwent a radioactive iodine treatment 7 years previously. She is also a heavy smoker. CT and PET/CT showed a 4 cm lesion at the left lower lobe (SUV-max 9.2 g/ml) with a suspicious diaphragma invasion. Left lower lobectomy performed with partial diaphragma resection. Pathological specimen was leiomyosarcoma grade-1, with pleura invasion, lymph nodes and diaphragmatic margins were tumor negative. Patient had received chemotherapy and radiotherapy, but she died after19 months due to the progression of the tumor.
Case 4
A 70-year-old man admitted to our clinic with hoarseness and chest pain. He was a heavy smoker and had a history of asbestosis. He had a family history of lung cancer. CT and PET/CT scans revealed a lesion at right vocal cord (SUV-max 7.1 g/ml), left intraclavicular and mediastinal multipl lymph nodes (SUV-max 14.5- 22.3 mg/dl) and a 4.1 cm solid lesion of left upper lobe (SUV-max 18.9 g/ml). Endobronchial ultrasound could not be performed because the patient did not accept any approach with local anesthesia or sedation. Broncoscopy was performed under general anesthesia and carcinosarcoma was diagnosed. He received chemotherapy and radiotherapy but died 2 months later with progression of the tumor.
Case 5
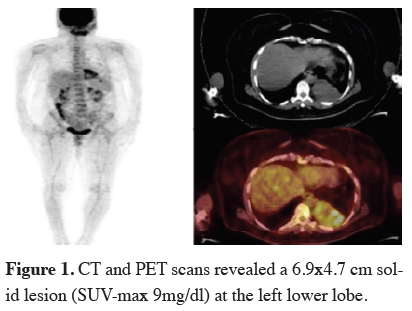
A 51-year-old women presented with a left lateral chest pain. She had a past medical history of tumor resection from her leg and received radiotherapy 30 years previously with the diagnosis of fibrosarcoma. She also had a family history of liver carcinoma, larynx carcinoma and lymphoma. At CT and PET/CT scans a 6.9x4.7 cm lesion (SUV-max 9mg/dl) at the left lower lobe was found (Figure 1). Left lower lobectomy with partial diaphragma resection was performed. Pathological specimen was synovial sarcoma with pleural and muscular invasion. There were no lymph node invasion and free margins of tumor. She was operated 1 month ago, chemotherapy and radiotherapy will be planned.
 Click Here to Zoom |
Figure 1: CT and PET scans revealed a 6.9x4.7 cm solid lesion (SUV-max 9mg/dl) at the left lower lobe. |
Discussion
Sarcomas are uncommon primary malignant tumors of the lung and account for less than 0.5% of the cancers of the lung so we have only a little experience about these tumors [2]. Cameron found only nine patients in a series of 6000 primary lung neoplasms (0.15%) in 1975, and Miller found only 30 out of 10134 lung neoplasms (0.3%) [2,3].Detailed IHC studies began after 1985 and then allowed a better classification of sarcomas, based on the predominant histological pattern of growth and cytological composition [1]. In the lung, primitive mesenchymal cells may arise from the stromal elements of the parenchyma like bronchial or vascular wall. They usually expand towards the parenchyma [4].
Parenchymal and endobronchial sarcomas include; fibrosarcoma, leiomyosarcoma, rhabdomyosarcoma, hemangiopericytoma, osteosarcoma, myxosarcoma, spindle cell sarcoma, angiosarcoma, malignant fibrous histiocytoma, malignant schwannoma, malignant mesenchymoma, undifferentiated sarcoma, pulmonary blastoma, neurogenic sarcoma, chondrosarcoma, synovial sarcoma and liposarcoma [1]. The most common types are leiomyosarcomas, fibrosarcomas and hemangiopericytomas. In this study, carcinosarcoma was the most common type.
Incidence of primary pulmonary sarcomas are equal in males and females, and can occur at any age with a range of 4 to 83 years showing a slight predominance at middle ages [1,2]. In this study, there are 3 male and 2 female patients between 51-70 years age.
Most of the patients are asymptomatic and the mass is incidentally diagnosed [1]. Only one patient in this series is asymptomatic who had a small tumor size (2.1 cm). Other patients having tumors of 4 to 7.5 cm had symptoms such as dyspnea, hoarseness, hemoptysis and chest pain. Fever, fatigue, anorexia and weight loss are later symptoms. Presenting symptoms are similiar to those of bronchogenic carcinoma. On radiographs the tumor is usually presented as a large peripheral well-circumscribed solitary mass and vary in diameter from 1 to 15 cm. In this series, it varied from 2.1 to 7.5 cm [1].
Thorax CT scan is an important diagnostic tool as the tumor often invades pleura, pericardium, vessels and chest wall [5]. Two patients in this series had diaphragmatic invasion, they were diagnosed as leiomyosarcoma and synovial sarcoma. The tumors were 4 and 6.9 cm, and left lower lobectomy performed for both of them with an additional partial diaphragma resection.
Differential diagnosis of primary lung sarcoma is critical for the treatment. The gross presentation of these tumors does not differ significally from the more common tumors of the lung and from the metastatic sarcomas. Detailed clinical history, presence of past soft tissue neoplasms, previous radiation exposure is critical for distinction of these tumors. The tumors should be diagnosed with histological methods and molecular techniques immediately after other alternative diagnosis were excluded. Small biopsy samples might be very challenging so wide resection materials are better for diagnosis. Treatment and prognosis differs from the other soft tissue sarcomas and bronchogenic carcinomas [2,6].
Complete surgical resection with lobectomy, pneumonectomy or segmentectomy with pathological free margins is the best option for treatment [4]. We performed 4 lobectomy in 5 patients. In most of the series in the literature, a high percentage of large tumors were reported and resections are extended to the chest wall, diaphragm, superior vena cava or auricle. Two patients in this series diaphragmatic resection was performed. Porte reported extended resections at 6 out of 18 patients and Bacha at 6 out of 19 patients. [4]. Mediastinal lymph node dissection was performed in all patients. Primary lung sarcomas rarely disseminate to lymph nodes. Regnard et al. found N2 involvement in 2 out of 20, and N1 involvement in three patients [4]. Mediastinal lymph node dissection were routinly performed at our clinic and we did not see any lymph node invasion except the patient that was diagnosed at the terminal stage.
Positive resection margins are related poor prognosis. Bacha reported 6 patients out of 20 had positive resection margins [4]. Large tumor size is also a poor prognostic factor. According to Nascimento et al. 5 cm and Jansen et al. 4 cm tumor size indicated a poor prognosis [4]. In the series of MacCormack and Martini, and Burt and Zakowski a tumor diameter of 5 cm or less is related better prognosis. However Regnard and Bacha did not find that tumor size a prognostic factor [4]. Additional chemoterapy and/or radiotherapy is a piece of advice of treatment but the efficacy of them is still poor because they do not have standart protochols and they need further studies [4].
Carcinosarcoma of lung is a rare tumor and its incidence is 0.1 to 0.4% of all lung malignancies [7]. In the literature, pulmonary carcinosarcoma was first described in 1908, that a complex tumor for diagnosis [8]. The World Health Organization (WHO) classification of 2015 identified carcinosarcoma as a subtype of sarcomatoid carcinoma and defined as malignant neoplasm composed of both malignant epithelial and mesenchymal elements [1,9]. The most frequent epitheliel component is squamous carcinoma and sarcomatous compenent is rhabdomyosarcoma. Our two patients also had the two components just like the literature [8,10]. In the literature, fewer than 20 cases are reported until 1995 [11-14]. Ishida et al. [15] reported 5 cases, Kimino et al. reported a case, Grahmann et al, Berho et al, Zehani et al. and Wick et al reported 3 cases of each [1,16]. Xu et al. [17] reported 48 cases and this is one of the largest series of the literature. They usually seen an average age of 60-year-old males of heavy smokers [8]. We have two male patients of 58 and 70 year ages and both of them were heavy smokers. Carcinosarcomas usually grow slowly and clinical symptoms depend on the location and size of the tumor. Complete resection of the tumor is the essential treatment. The role of chemotherapy is still very poor. Liang et al. [18] and Raveglia et al. [19] both reported failures of patients’ survival. [7] The five year survival rate varies between 6-49.4% [1,8]. According to Xu et al. [17] the 1-3 and 5-year survival rate was 77.1%, 49.5% and 22.7%. Our one patient is still alive for 52 months and free of disease, the other patient died within two months, but he was in the terminal level when diagnosed.
Undifferentiated pleomorphic arcoma, redefined by Fletcher in 2002, after the development of IHC techniques they examined more carefully. Before IHC, they diagnosed the cases as malignant fibrous histiocytoma [1,20]. They are rare tumors, 0.1-0.4% of all lung malignancies [21]. They are high grade sarcomas and show progressive and rapid growth. Sarcomas have an aggresive clinical course, metastasis and recurrence incidance is high [22]. Surgical resection is gold standart for treatment, and we performed lobectomy to our patient. Palliative chemotherapy or radiotherapy may be used. Our patient had a small tumor of 2.1 cm diamater and there were no lymph node invasion, surgical margins were free of tumor, so he did not take chemoradiotherapy. 5-year survival of undifferentiated pleomorphic sarcoma is approximately 50% [22]. During the follow up of 33 months he had no metastasis or recurrence. Xu reported a 59-year-old woman with symptomatic right atrium metastasis of an undifferentiated pleomorphic sarcoma of lung. She diagnosed as stage 4 lung cancer of left upper lobe and she has undergone chemoradiotherapy. After a followed up of 4 years, she had been operated for atrial metastasis and follow up for two months without recurrence [22]. Goncalves et al [23] reported a 66-year-old man, diagnosed as undifferentiated pleomorphic sarcoma at the right upper lobe, he was treated with endobronchial tumor debulking by laser therapy and radiotherapy and survived over 11 years. Abreu et al [21] also reported a 74-year-old patient with a 7 cm lesion at right hemithorax.
Leiomyosarcoma, rarely arises in the lung, differantiated from smooth muscle and originated from pulmonary artery, bronchus or pulmonary parenchyma [6,24,25]. The tumor accounts 0.5% of the malignant tumors of lung [24]. Age range is wide, 5 to 76 years and our patient was 52-year-old. Most of the patients were asymptomatic and if symptom is present they are usually chest pain, hemoptysis and cough. The tumor can present with Pancoast tumor [25]. Our patient referred to our clinic with dyspnea and hemoptysis. The radiological features are nonspecific, tumor sizes varies from 1.7 to 10 cm diameter. Our patient had a 4 cm lesion at the left lower lobe with suspicious diaphragma invasion. [26] The earliest case was reported in 1907 by Davidsohn [24,27]. Agnos and Starkey recorded 18 cases in 1958, Dyson and Tretalance reported 26 cases in 1964 [28] and Guccion et al and Nascimento et al have reported 41 cases [1]. Porte et al [4] reported 2 cases, Janssen et al [27] 8 cases and Mastroianni et al [2] 7 cases. Surgical resection is the best treatment option and lobectomy was performed with diaphragma resection. The surgical margins and lymph nodes were free of the tumor in our case. The tumor can make distance metastasis but it’s uncommon, Young et al reported the thyroid metastasis after lobectomy [24,29]. If an early and complete resection was performed, 5-year survival rate is reported as 50%. However the prognosis is usually poor, only a few patients lived 15 to 20 years, our patient died after 19 months from the progression of the tumor [24].
Synovial sarcoma, is defined by the 2004 WHO classification and it is a mesenchymal spindle cell tumor which variably displays areas of cellular differentiation [6,30,31]. Cytogenetic studies showed a translocalation over chromosome 18 [32]. Histopathologically its divided as; biphasic, monophasic fibrous, monophasic epithelial, and poorly differantiated types. Our patient was diagnosed as biphasic synovial sarcoma [32,33]. It is a rare tumor and only 0.1% of all resected malignant lung tumors, also 5-10% of all human soft tissue sarcomas [34-36]. They usually occurs in extremity, especially around the knee [37-39]. It is range is 3 to 84 years of age and slightly more frequent in woman [34]. The usual symptom is dyspnea, cough, chest pain, and hemoptysis similiar to the other tumors of the lung. The tumor was not encapsulated in at least 75% of reported cases just like our patient [6]. Niwa et al [40] reported a case that exhibits a polypoid endobronchial growth. Tumor size ranges between 0.6 to 17 cm and in our case it was 6.9 cm. Primary resection is the best treatment and lobectomy was performed in our patient. These tumors may infiltrate pleura, chest wall and mediastinum. Our patient had an invasion of diaphragma, pleura and muscular layer. Distance metastasis can also be seen. Ezzine et al reported an intracardiac metastasis, Ogino et al. brain metastasis and Seyhan et al vertebra metastasis [35,36,41]. Zeren et al reported 25 similar cases, Essary et al reported 12 cases [1,42]. Jiang et al reported approximately 73 cases reported in the literature since 2008, Nakano et al. reported 60 cases since 2010 and Ozdil et al. reported 90 cases since 2018 [31,33,38,39]. The patient is in the first month of follow up, chemotherapy and radiotherapy is planned. The tumor is chemosensitive, adriamycin alone or in combination with ifosfamide remains the standart protochol. Radiotherapy is used for achieving excellent local control after surgery [37]. Metastasis and recurrence rates are low so 10-year survival can be seen, survival rates are between 1 month to 19 years [38]. High grade metastatic tumors are with poor diagnosis and 75% dead within two years. Poor prognostic factors are, tumor size over 5 cm, male sex, greater than 20 years of age, extensive tumor necrosis, neurovascular invasion and large number of mitotic figures [30]. Generally 5 year survival rate is 30-50% [30,37]. Spontan regression of tumor without a treatment is also very rare and occur in 1 in 60,000 to 100,000 cases of all malignancies [43]. Tsunezuka et al reported a 38-year-old patient with spontaneous regression of tumor after transbronchial biopsi, 2 months after biopsi lobectomy performed to the patient but there were no tumor at the pathological examination [43].
In conclusion primary sarcomas of the lung are very rare and aggresive malignancies. Clinically they cannot be differentieted from more common tumors of the lung and metastatic sarcomatous tumors. Early diagnosis and complete surgical resection before the tumor become larger and metastatical is critical for prognosis. Efficacy of chemotherapy and/or radiotherapy is still not clear so a multidisciplinary approach may be useful. Primary sarcomas of the lung must always been remembered for the differantial diagnosis of the other tumors and a complete resection should be performed immediately for better prognosis.
Declaration of conflicting interests
The authors declared no conflicts of interest with respect to the authorship and/or publication of this article.
Funding
The authors received no financial support.
Reference
1) Shields TW, Robinson PG. Uncommon primary malignant tumors of the lung. In: Shields TW, Locicero J, editors. General Thoracic Surgery. Philadelphia: Lippincott Williams&Wilkins; Seventh edition, 2009. p. 1591-618.
2) Mastroianni BE, Falchero L, Chalabreysse L, Loire R, Ranchère D, Souquet PJ, et al. Primary sarcomas of the lung. A clinicopathologic study of 12 cases. Lung Cancer 2002; 38: 283-9.
3) Cameron EWJ. Primary sarcoma of the lung. Thorax 1975: 30: 516.
4) Porte HL, Metois DG, Leroy X, Conti M, Gosselin B, Wurtz A. Surgical treatment of primary sarcoma of the lung. Eur J Cardiothorac Surg 2000; 18: 136-42.
5) Çakır Ö, Topal U, Bayram AS, Tolunay Ş. Sarcomas: rare primary malignant tumors of the thorax. Diagn Interv Radiol 2005; 11: 23-7.
6) Litzky LA. Pulmonary sarcomatous tumors. Arch Pathol Lab Med 2008; 132: 1104-17.
7) Kanzaki R, Ikeda N, Okura E, Kitahara N, Okimura A, Kawahara K, et al. Pulmonary carcinosarcoma with an osteosarcomatous component. Gen Thorac Cardiovasc Surg 2012; 60: 855-8.
8) Gleason T, Haas M, Le BH. Imaging, Histopathologic, and Treatment Nuances of Pulmonary Carcinosarcoma. Case Reports Radiol 2017; Article ID 8135957, 5 pages.
9) Yang H, Lin Y, Liang Y. Treatment of lung carcinosarcoma and other rare histologic subtypes of non-small cell lung cancer. Curr Treat Options in Oncol 2017; 18: 54.
10) Citil R, Cıralı H, Karslıgil A. Carcinosarcoma of the Lung Associated with Neurofibromatosis Type 1: A Case Report. Turk J Pathol 2012; 28: 90-4.
11) Suster S. Primary sarcomas of the lung. Semin Diagn Pathol 1995; 12: 140-57.
12) Colby TV, Bilbao JE, Battifora H, Unni KK. Primary osteosarcoma of the lung: a reappraisal following immunohistologic study. Arch Pathol Lab Med 1989; 113: 1147-50.
13) Chapman AD, Pritchard SC, Yap WW, Rooney PH, Cockburn JS, Hutcheon AW, et al. Primary pulmonary osteosarcoma: case report and molecular analysis. Cancer 2001; 91: 779-84.
14) Krygier G, Amado A, Salisbury S, Fernandez I, Maedo N, Vazquez Maedo N, et al. Primary lung liposarcoma. Lung Cancer 1997; 17: 271-5.
15) Ishıda T, Tateishi M, Kaneko S, Yano T, Mitsudomi T, Sugimachi K, et al. Carcinosarcoma and spindle cellcarcinoma of the lung. Clinicopathologic and immunohistochemical studies. J Thorac Cardiovasc Surg 1990; 100: 844.
16) Zehani A, Ayadi-Kaddour A, Mlika M, Fkih L, Marghli A, Fenniche S, et al. Le carcinosarcome pulmonaire. La tunisie Medicale 2013; 91: 287-9.
17) Xu Q, Zhou C, Ni J, Zhou S, Jiang G, Ding J, et al. Clinical characteristics and prognosis analysis of pulmonary carcinosarcoma (a report of 48 cases). Chinese J Lung Canc 2007; 10: 148-51.
18) Lin Y, Yang H, Cai Q, Wang D, Rao H, Lin S, et al. Characteristics and prognostic analysis of 69 patients with pulmonary sarcomatoid carcinoma. Am J Clin Oncol 2016; 39: 215-22.
19) Raveglia F, Mezzetti M, Panigalli T, Furia S, Giuliani L, Conforti S, et al. Personal experience in surgical management of pulmonary pleomorphic carcinoma. Ann Thorac Surg 2004; 78: 1742-7.
20) Fletcher CDM, Unni KK, Mertens F. World Health Organization classification of tumours, pathology and genetics of tumours of soft tissue and bone. Lyon; IRC 2002; 10.
21) Abreu MM, Kozlowski BM, Cordeiro PB, Sousa A, F Nascimento M, Zamboni M. Primary pleomorphic sarcoma of the lung. Rev Port Pneumol 2008; 4: 535-9.
22) Xu G, Shi X, Shao G. An unusual case of metastasis of a pulmonary undifferentiated pleomorphic sarcoma to the right ventricle: a case report. J Med Case Reports 2013; 7: 165.
23) Gonçalves MJ, Mendes MM, João F, Lopes JM, Honavar M. Primary pleomorphic sarcoma of lung 11 year survival. Rev Port Pneumol 2011; 17: 44-7.
24) Shen W, Chen J, Wei S, Wang X, Li X, Zhou Q. Primary pulmonary leiomyosarcoma. Chinese Med Assoc 2014; 77: 49-51.
25) Shojia F, Yoshinoa I, Takeshitab M, Sumiyoshib S, Sueishib K, Maeharaa Y. Pulmonary leiomyosarcoma presenting as a pancoast tumor. Pathology – Research and Practice 2003; 2007: 745-8.
26) Hayashi T, Tagawa T, Ashizawa K, Abe K, Kinoshita N, Tsutsui S, et al. Contrast-enhanced dynamic magnetic resonance imaging of primary pulmonary leiomyosarcoma. Tohoku J Exp Med 2006; 210: 263-7.
27) Janssen JP, Mulder JJS, Wagenaar SS, Elbers HR, van den Bosch JM. Primary sarcoma of the lung: a clinical study with long-term follow-up. Ann Thorac Surg 1994; 58: 1151-5.
28) Ramanathan T. Primary leiomyosarcoma of the lung. Thorax 1974; 29: 482.
29) Young KW, Ranb KY, Uka WS, Boka LJ. Pulmonary leiomyosarcoma metastatic to the thyroid gland: Case report and review of the literature. Ann d’Endocrinol 2011; 72: 314-6.
30) Bhattacharya D, Datta S, Das A, Halder KC, Chattopadhyay S. Primary pulmonary synovial sarcoma: A case report and review of literature. In J App Basic Med Res 2016; 6: 63-5.
31) Özdil A, Bozdağ HG, Yaman B, Çakan A, Çağırıcı U. An extremely rare lung tumor of a young adult: Primary synovial sarcoma. Turkish J Thorac Cardiovasc Surg 2018; 26: 157-60.
32) Park JS, Min BR, Park SH, Kwon KY, Keum DY, Choi W. Primary Pulmonary Biphasic Synovial Sarcoma Confirmed by Molecular Detection of a SYT-SSX2 Fusion Gene: Report of 1 Case. Korean J Intern Med 2010; 25: 331-6.
33) Nakano J, Yokomise H, Huang CL, Misaki N, Chang SS, Okuda M, et al. Progressive growth of primary synovial sarcoma of the lung. Ann Thorac Cardiovasc Surg 2010; 16: 194-7.
34) Ichimuraa H, Kikuchia S, Ozawaa Y, Matsuzakib K. Primary synovial sarcoma of the lung successfully resected under temporary bypass. Interact Cardiovasc Thorac Surg 2013; 17: 588-90.
35) Ogino H, Hanibuchi M, Takizawa H, Sakiyama S, Sumitomo H, Iwamoto S, et al. Primary Pulmonary Synovial Sarcoma Showing a Prolonged Survival with Multimodality Therapy. Int Med 2016; 55: 381-7.
36) Seyhan EC, Sokucu SN, Gunluoglu G, Veske NS, Altin S. Primary Pulmonary Synovial Sarcoma: A Very Rare Presentation.Case Rep Pulmonol 2014; 2014: 537618.
37) Panigrahi MK, Pradhan G, Sahoo N, Mishra P, Patra S, Mohapatra PR. Primary pulmonary synovial sarcoma: A reappraisal. J Can Res Ther 2018; 14: 481-9.
38) Jiang J, Zhou J, Ding W. Primary pulmonary synovial sarcoma, a rare primary lung neoplasm: Two case reports and reviewof the current literature. Respirology 2008; 13: 748-50.
39) Hosono T, Hironaka M, Kobayashi A, Yamasawa H, Bando M, Ohno S, et al. Primary pulmonary synovial sarcoma confirmed by molecular detection of SYT-SSX1 fusion gene transcripts: a case report and review of the literature. Jpn J Clin Oncol 2005; 35: 274-9.
40) Niwa H, Masuda S, Kobayashi C, Oda Y. Pulmonary synovial sarcoma with polypoid endobronchial growth: a case report, immunohistochemical and cytogenetic study. Pathol Int 2004; 54: 611-5.
41) Ezzine SB, Chelbi E, Bouzaidi K. Intracardiac metastasis of primary synovial sarcoma of the lung. Asian Cardiovasc Thorac Ann 2012; 21: 623.






